–
Apoptosis
Will you offer us a hand? Every gift, regardless of size, fuels our future.
Your critical contribution enables us to maintain our independence from shareholders or wealthy owners, allowing us to keep up reporting without bias. It means we can continue to make Jewish Business News available to everyone.
You can support us for as little as $1 via PayPal at office@jewishbusinessnews.com.
Thank you.
–
/By Alan Gallindoss /
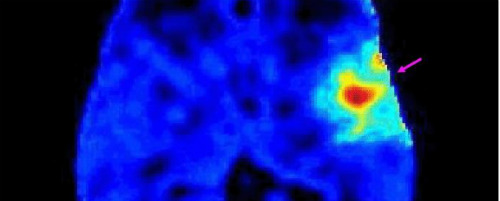
Apotex has developed new molecular-level imaging technology to detect diseases within human cells, and potentially help therapeutically target cells that are cancerous directly, without harming healthy ones. The company’s share price responded strongly this morning to the new patent announcement, before falling back again to give up half of its gains, after Aposense formally announced it has obtained a US patent on its ATT-11T molecule for the treatment of colon, lung, and ovarian cancers. The patent covers the molecule’s assembly, and will expire on in seventeen years time on November 22, 2030.
Many diseases begin in the same way, with a process of cell death – known as “apoptosis”. Aposense has developed a molecular imaging technology to detect apoptosis directly in cells at the earliest stage of disease. The molecule ATT-11T was developed by Aposense together with Teva Pharmaceutical Industries Ltd.
The new molecule is a conjugate of the active ingredient in irinotecan (SN-38), a well-known cancer drug, combined with proprietary Aposense molecules which together extend irinotecan’s effectiveness and reduce its toxicity. ATT-11T has been found in pre-clinical trials to have superior efficacy than irinotecan by itself in terms of the amount of the active ingredient in the tumor tissue and longer time in the bloodstream, imparting it an edge over current cancer drugs by extending the effectiveness of the treatment.
According to the company Aposense technology is broadly also potentially applicable to many diseases, whether they are inflammatory, cardiovascular, neurodegenerative, infectious or genetic. The diagnostic technology of Aposense can provide clinicians with quick and early treatment results. One of the biggest problems in oncology is that it can take months to identify a response, to see if a particular cancer treatment is working. Molecular imaging can show the response very early – within a day or week. This therefore can open a new approach for tailoring treatments to individual patients, within the framework of personalized medicine.
An additional benefit of molecular imaging diagnostics is its non-invasive approach. It works by injecting the molecules into cancerous cells with a therapeutic compound. This approach increases effectiveness, while dramatically reducing dosage and side effects of chemotherapy. Aposense claims that by linking it with a probe it can become a specifically targeted treatment, and may achieve a superior result using only 20% of the current dose.
Aposense CEO Dr. Miri Ben-Ami said today, “Approval of the patent is an important milestone for Aposense, in view of the company’s progress in therapeutics, where ATT-11T is our flagship product. We plan to file a pre-investigational new drug (IND) with the US Food and Drug Administration (FDA) for further development of the drug, including clinical trials. The company’s patent strategy is intended to provide multi-layered patent protection for its products. We believe that the patent which was approved is proof of the company’s patent standing and its proprietary platform.”
The patent news may best perhaps be summed up as partial good news. The shares of Aposense have indeed had a wild ride since the heady days of their IPO almost three years ago, when the shares debuted at a price more than ten times higher than they are today. Just last December even, one of their molecules emerged from Phase 2 clinical trials with inconclusive results which led the share price down to recent levels of one quarter of where they traded then. Still, while progress in bio-tech can certainly take a long time, for those who are patient the rewards may yet justify the hardship of waiting and the company’s sponsorship remains strong.