–
Protalix BioTherapeutics announced today, June 19th, 2013, that it has entered into a supply and technology transfer agreement with Fundação Oswaldo Cruz (Fiocruz), an arm of Brazil’s Ministry of Health, for UPLYSO™ (“taliglucerase alfa“), the Company’s proprietary enzyme replacement therapy for the treatment of Gaucher disease. Gaucher disease is a rare lysosomal storage disorder that affects approximately 10, 000 people worldwide, and which can occur, particularly, too in Jews of Ashkenazi origin.
The technology transfer is expected to take place during the initial seven-year term of the agreement, and is intended to transfer to Fiocruz the capacity and skills required for the Brazilian Government to construct its own manufacturing facility, at its sole expense, and to produce a sustainable, high quality, and cost effective supply of UPLYSO. Under the agreement, Fiocruz has committed to purchase at least approximately $40 million worth of UPLYSO from Protalix during the first two years of the agreement.
In subsequent years, Fiocruz is required to purchase at least approximately$40 million worth of UPLYSO per year. Additionally, Protalix is not required to complete the final stage of the technology transfer until Fiocruz purchases at least approximately US$280 million worth of UPLYSO in total. The agreement may be extended for an additional five-year term, as needed, to complete the technology transfer. All of the terms of the initial arrangements, including minimum annual purchases, will also apply during an additional term.
Protalix CEO Dr. David Aviezer said, “We are excited to be working with the Brazilian government in its efforts to provide Uplyso to Gaucher patients in Brazil and to collaborate with Fiocruz regarding our plant cell technology. We are encouraged by the recognition of our drug and our technology by both Fiocruz and Brazil’s Ministry of Health, and believe that this agreement will further establish a reliable supply of treatment for patients living with Gaucher disease.”
Brazilian Minister of Health Dr. Alexandre Padilha said, “Through this collaboration, we are able to strengthen our technological and industrial capabilities in the area of biologics manufacturing and improve the health of Brazilian citizens who are impacted by this rare disorder.”
In March 2013, the Brazilian National Health Surveillance Agency granted regulatory approval to Pfizer for Uplyso (which initially had licensed marketing rights for Brazil from Protalix), which is indicated for the long-term enzyme replacement therapy for adults with a confirmed diagnosis of Type I Gaucher disease.
Will you offer us a hand? Every gift, regardless of size, fuels our future.
Your critical contribution enables us to maintain our independence from shareholders or wealthy owners, allowing us to keep up reporting without bias. It means we can continue to make Jewish Business News available to everyone.
You can support us for as little as $1 via PayPal at office@jewishbusinessnews.com.
Thank you.
UPLYSO is marketed as ELELYSOTM in the United States and Israel. To facilitate the arrangement with Fiocruz, Protalix’s commercialization partner for Uplyso, Pfizer Inc., has amended its exclusive license and supply agreement with Protalix and returned commercialization rights in Brazil to the company. In consideration for the return of these rights, Protalix will pay Pfizer a maximum amount of $12.5 million from the Company’s net profits generated in Brazil for each year of the agreement. During the return of commercial rights back to Protalix, Pfizer will continue to support Gaucher disease patients in Brazil who are being treated with UPLYSO.
Protalix is holding a previously scheduled analyst event on Thursday, June 20, 2013 at 8:00 AM EDT, and management will elaborate there on certain of the terms and conditions of the new agreement during its presentations. Audio of the event and the slide presentation will be webcast live and archived on the Company’s website for a 30-day period.
About Protalix
Protalix BioTherapeutics is an Israeli pharmaceutical company whose plant-based enzyme, taliglucerase alfa, has been approved by the U.S. Food and Drug Administration for the treatment of Gaucher disease.
The company was founded in 1993 by Yoseph Shaaltiel, who received his Ph.D. in Plant Biochemistry from the Weizmann Institute in Israel and served in the Biology Department of the Israel Defense Forces’ Biological and Chemical Center. One of the earliest and largest investors in the company initially was Phillip Frost though he has since substantially reduced his holdings.
Protalix entered into a partnership agreement with Teva Pharmaceuticals Industries in 2006 for the development of two proteins and in 2009 signed a collaboration agreement with Pfizer for the development and commercialization of its taliglucerase alfa treatment. Also in 2009, Protalix reported that Frost & Sullivan presented the company with its 2009 European Orphan Diseases (i.e. affecting fewer than 200, 000 sufferers) Market Product Innovation of the Year Award. In 2011 Protalix announced that the U.S. Food and Drug Administration had approved the company’s manufacturing facility in Carmiel.
Protalix uses cultured plant cells to manufacture its bio pharmaceuticals. As of 2012, it has developed two such
products, the second of which continues to undergo clinical trials:
Taliglucerase alfa – a recombinant glucocerebrosidase enzyme produced from transgenic carrot cell cultures. Known also as Elelyso, taliglucerase won approval from the U.S. Food and Drug Administration in May 2012 as an orphan drug for the treatment of Type 1 Gaucher’s Disease.
PRX-105 – a recombinant human Acetylcholinesterase, also produced from transgenic carrot cells which can be used as a counter-measure against nerve agent attacks. PRX-105 has so far completed Phase I clinical trails.
Protalix initially became a public company through a reverse takeover of Orthodontix, a company which was at the time traded over the counter on NASDAQ. This merger was completed on December 31, 2006, which at the time valued the new joint entity at almost US$1 Billion. The company subsequently applied for a listing on the AMEX, and sold 10 million shares in a public offering.
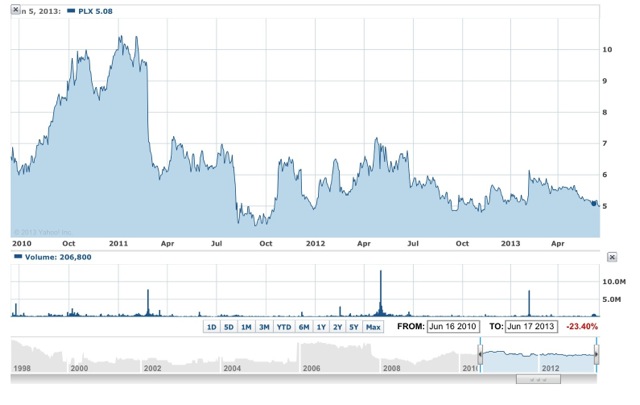
Today, its common shares currently trade with a market cap less than half that total valuation, – about US$470 million on June 19th, 2013 on the NYSE MKT exchange for small and medium cap companies, at a price of around US$5.08 per share. Only, now with many more – 93, 502, 230 – shares outstanding (as at March 31st, 2013) after a number of public and privately placed preferred and common share offerings since the original merger. The share price has also had a number of ups and downs in the years since following trends in the market itself, as you can see in the chart for the last three years alone:
–
Chart courtesy of Yahoo Finance
–
So the bio-tech road can be a rocky one even for companies with excellent prospects such as Protalix. At its current burn-rate however Protalix need not worry too much – with almost US$42 million in the bank they have lots of time to develop the business, as well as some well-heeled investors in the background. With a successful analysts presentation on June 20th investors should have a good understanding of the progress the company is now making.