–
Brainsway CEO Uzi Sofer
Will you offer us a hand? Every gift, regardless of size, fuels our future.
Your critical contribution enables us to maintain our independence from shareholders or wealthy owners, allowing us to keep up reporting without bias. It means we can continue to make Jewish Business News available to everyone.
You can support us for as little as $1 via PayPal at office@jewishbusinessnews.com.
Thank you.
/By Alan Gallindoss /
Brainsway Ltd., the developer of a magnetic device for treating neurological conditions, reported today June 19th, 2013 in a Tel Aviv Stock Exchange filing that a U.S. company will help the Israeli manufacturer market the product in the United States.
According to Bloomberg, the so-far undisclosed partner will be allowed to approach certain potential customers of the Brainsway “Deep TMS” system, as specified by Brainsway in the agreement. Brainsway will also continue to market the device to other U.S. customers directly and already has “some direct-lease agreements, ” Brainsway CEO Uzi Sofer said.
The U.S. Food and Drug Administration and Canadian health regulator Health Canada approved Deep TMS in January for use on patients who fail to respond to, or can’t tolerate, antidepressants. Sofer said in an interview at the time that a marketing accord was planned in the first quarter, possibly including a “large” up-front payment. Brainsway did not outline any financial terms in today’s filing.
The marketing agreement “is a part of our wider effort to focus on expanding our activities in the United States”, Sofer said in a telephone interview with Bloomberg, “We will simultaneously continue with our own direct sales efforts to U.S. customers.”
The new marketing partner will work to lease about 200 devices over as many as three years for use in the United States, Brainsway said. Sofer declined to specify any combined targets for his company and the new U.S. marketer, why the agreement isn’t nationwide or whether Brainsway is in talks with other potential partners.
According to the company’s website Brainsway’s patented breakthrough technology launches a new era in brain disorder treatment. A non-invasive, yet highly effective solution, Brainsway can make a difference in the wellbeing and health of millions of patients worldwide the company claims.
Brainsway’s technology releases millions of patients from a bleak cycle of insufficiently effective treatments and unpleasant side effects the corporate website states. Brainsway’s high efficacy for the treatment of Major Depressive Disorder is now validated for use by the FDA, which has granted a wide indication for the treatment of Major Depressive Disorder patients. The company reports the technology is also safe, with no systemic effects or long-term side effects, and also highly convenient, requiring only brief daily sessions over a couple of weeks.
Brainsway reports it has has also gained CE approval – for the European Union – for the following indications:
Major Depressive Disorder (FDA indication)
Bipolar Depression
Post-Traumatic Stress Disorder
Schizophrenia (negative symptoms)
Parkinson’s Disease
Chronic Pain
The technology has apparently already been applied in thousands of cases worldwide in the framework of clinical studies. The company states it has also been enthusiastically embraced by leading research institutions worldwide, with over 60 clinical trials performed.
–
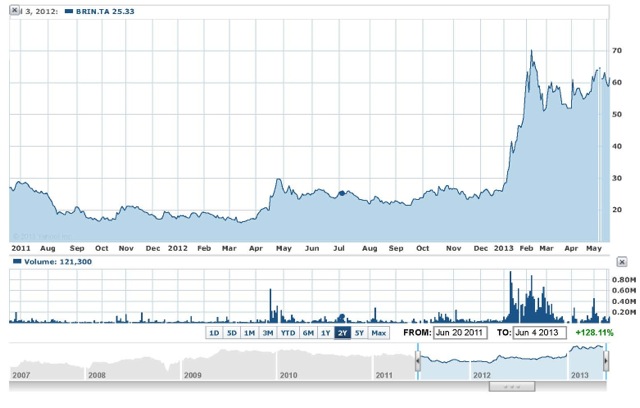
Brainsway Stock Rises
Chart: courtesy of Yahoo Finance
Brainsway’s shares rose by 7 percent to almost 60 shekels today, the biggest intra-day gain it has experienced since May 1, and was still trading up 2.7 percent by 4:30 p.m. in Tel Aviv, valuing the company at 732 million shekels or about US $204 million. The stock, which has fallen about 20 percent from its record high of almost 72 shekels on Feb. 10, has still more than doubled since the beginning of 2013.
The Deep TMS System uses a coil placed against the patient’s head to apply brief magnetic pulses to the brain, a process known as transcranial magnetic stimulation. Patients are treated five times a week for four weeks, with each session lasting about 20 minutes.
The company is counting on the technology’s appeal to psychiatrists and patients as a treatment for depression that eliminates the need for surgery or pharmaceuticals – some of which may need to be taken for life.
The TMS device also received CE European Union product-safety clearance in late May for treating Alzheimer’s disease and autism, as well as for use by people trying to give up cigarette smoking.
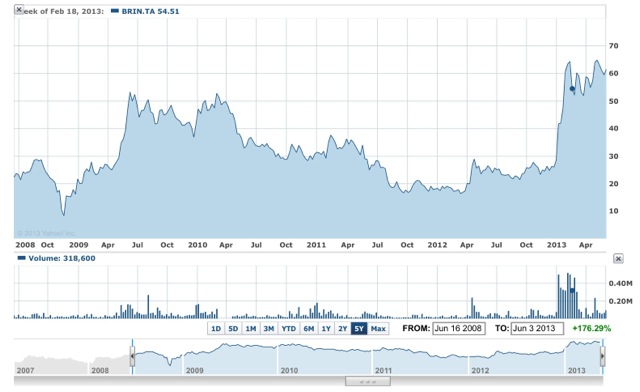
It has certainly been a long road traveled for Brainsway. A five year look at their stock chart shows a more complicated story:
–
Chart: courtesy of Yahoo Finance
–
So Brainsway’s shares last breached the $50 shekel mark three years ago, early in 2010 before falling back again. Nevertheless if, when the full details are known, nothing adverse should be in the fine print then it looks quite positive for the company.